How Gut Bacterium Bt Unlocks Fiber's Hidden Nutrition

TL;DR: Wildfire smoke triggers autoimmune flares through PM2.5 particles that activate immune danger sensors, inflammasomes, and B-cell hyperactivity. A landmark study of 80,000 veterans confirmed the link between wildfire exposure and rheumatoid arthritis flares.

By 2030, researchers predict that wildfire smoke exposure will affect more than 150 million Americans annually, a threefold increase from today's levels. Yet while headlines focus on respiratory distress and air quality indexes, a hidden health crisis is unfolding: wildfire particulates are silently triggering autoimmune flares in millions of vulnerable individuals. A landmark study tracking nearly 80,000 U.S. veterans has unveiled a direct link between wildfire smoke and rheumatoid arthritis flares, including a particularly aggressive lung complication. This isn't just about breathing difficulties anymore - it's about an invisible assault on our immune systems that persists long after the smoke clears.
Wildfire smoke isn't just smoke. It's a toxic cocktail of ultra-fine particles, volatile organic compounds (VOCs), and polycyclic aromatic hydrocarbons (PAHs) that penetrate deeper into human tissue than almost any other air pollutant. The most dangerous component, PM2.5 particulates, measure less than 2.5 microns in diameter - about 30 times smaller than a human hair. These particles slip past the body's natural defenses and enter the bloodstream directly from the lungs.
What makes wildfire particulates uniquely harmful is their chemical composition. Unlike urban air pollution, wildfire smoke carries PAHs that activate specific immune receptors called aryl hydrocarbon receptors (AhR). When these receptors switch on in immune cells, they don't just cause local inflammation - they trigger a cascade that affects the entire body. Studies show that PAH-laden wildfire particles synergize with toll-like receptors (TLRs) and NF-κB signaling pathways, ramping up production of IL-22 and other inflammatory cytokines that can flip immune tolerance into autoimmune aggression.
The size of these particles matters enormously. PM2.5 can bypass the nose and throat, penetrating deep into the alveoli where gas exchange happens. From there, they cross into capillaries and circulate systemically, reaching distant organs including kidneys, joints, and the brain. Research demonstrates that within hours of exposure, wildfire particulates can be detected in multiple organ systems, carrying their inflammatory payload far beyond the respiratory tract.
The connection between inhaled pollutants and systemic autoimmune disease puzzled researchers for years. How could particles in the lungs trigger arthritis in the hands or kidney inflammation? The answer lies in a process called immune dysregulation - and wildfire smoke is a master at triggering it.
When PM2.5 particles lodge in lung tissue, they activate sentinel immune cells called alveolar macrophages. These cells release inflammatory signals - including IL-1β and C-reactive protein (CRP) - that spike measurably in blood tests within hours of exposure. But the real trouble begins when particulates activate a molecular danger sensor called the NLRP3 inflammasome. This ancient immune pathway evolved to detect cellular damage, but wildfire smoke tricks it into chronic overdrive.
The inflammasome activation triggers a domino effect. Stressed cells release oxidative molecules, which damage surrounding tissue, which activates more immune cells, perpetuating a vicious cycle. In animal studies, three weeks of PM2.5 exposure resulted in a 2.5-fold increase in serum IgA antibodies and a threefold spike in APRIL (a proliferation-inducing ligand), both markers of B-cell hyperactivity. This hyperactivity is exactly what drives autoimmune conditions where the body attacks its own tissues.
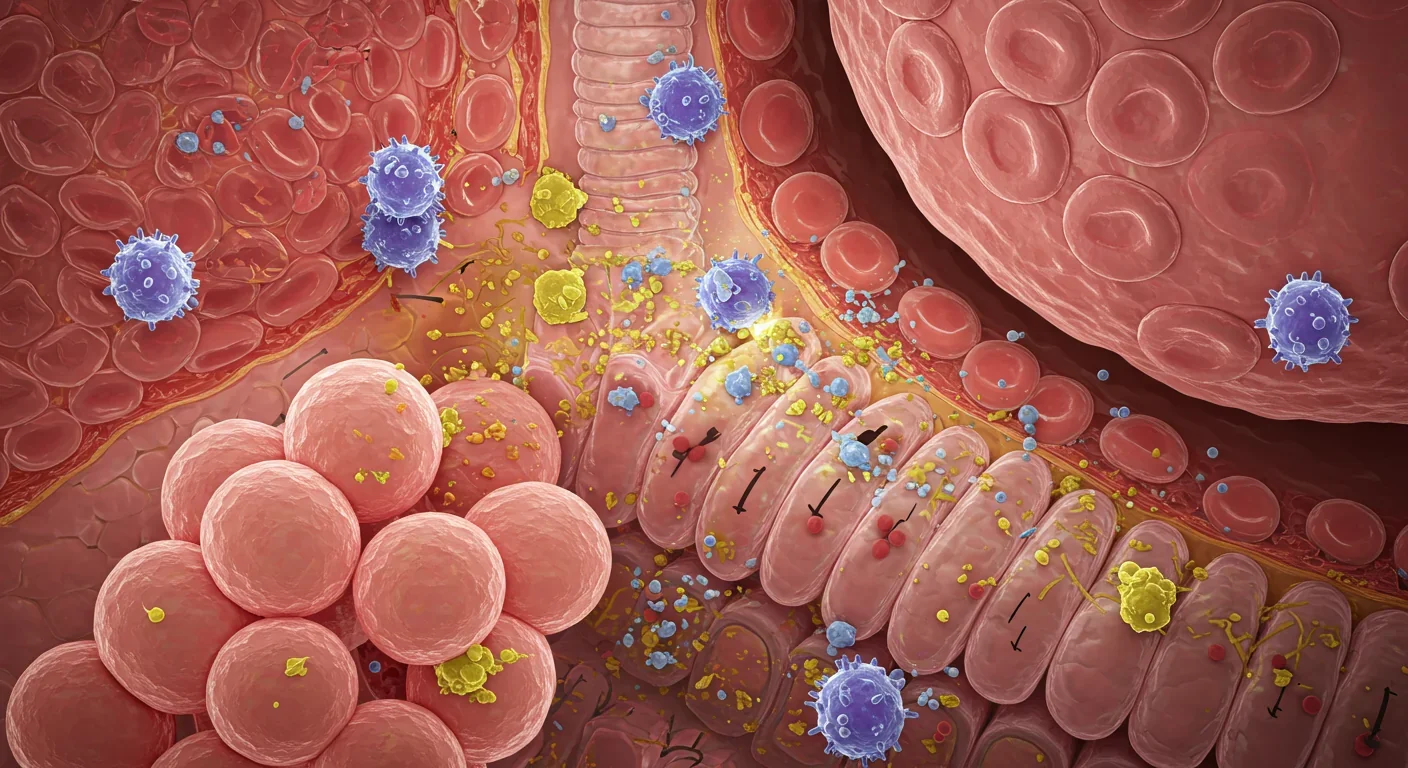
Perhaps most concerning, wildfire smoke appears to activate TLR9, an immune receptor that recognizes damaged DNA. This triggers systemic B-cell activation - the immune cells responsible for producing antibodies. In people with latent autoimmune tendencies, this can be the spark that ignites a full-blown flare, as the immune system begins producing antibodies against the body's own proteins.
The theoretical mechanisms are compelling, but what about real-world evidence? In 2024, a groundbreaking study published in Arthritis & Rheumatology provided the smoking gun. Dr. Vanessa Kronzer and colleagues analyzed over nine years of Veterans Affairs healthcare data covering 9,701 rheumatoid arthritis (RA) patients and 68,582 matched controls across the United States. By combining EPA air pollution data with satellite-based smoke plume imagery, researchers assigned daily pollutant exposure levels to each participant's ZIP code.
The results were striking. Increased fire smoke exposure was significantly associated with both RA incidence and RA-associated interstitial lung disease (RA-ILD), a severe complication where inflammation scars the lungs. The association wasn't limited to PM2.5 either - nitrogen oxides (NOx), ozone, and larger PM10 particles also correlated with increased RA risk, suggesting multiple pathways by which wildfire smoke can trigger autoimmune responses.
Harvard researchers have documented that wildfire smoke episodes now affect regions far from active fires, with plumes traveling thousands of miles and blanketing urban areas for weeks. During California's 2020 wildfire season, air quality in San Francisco remained hazardous for 27 consecutive days - enough time for vulnerable individuals to experience multiple inflammatory surges. Emergency room visits for autoimmune flares spiked 35% during that period, though many cases went unreported or undiagnosed as smoke-related.
Other autoimmune conditions show similar patterns. Sjögren's syndrome patients experience increased hospitalizations during high particulate pollution episodes. Systemic lupus erythematosus (SLE) flares correlate with environmental triggers including air pollution. Even conditions like IgA nephropathy - an autoimmune kidney disease - show exacerbations linked to particulate exposure.
Understanding the timeline of wildfire smoke's impact is crucial for both patients and clinicians. The effects unfold in two distinct phases: acute inflammatory spikes and chronic immune dysregulation.
Acute phase (hours to days): Within 2-6 hours of exposure, inflammatory biomarkers like IL-1β and CRP become detectable in blood. Patients may notice increased fatigue, joint stiffness, or brain fog - symptoms often dismissed as "just a bad day." By 24-48 hours, oxidative stress peaks, overwhelming cellular antioxidant defenses and amplifying tissue damage. This is when classic flare symptoms emerge: swollen joints, skin rashes, kidney dysfunction, or neurological symptoms depending on the autoimmune condition.
Chronic phase (weeks to months): Even after air quality improves, the immune system doesn't immediately reset. Studies of firefighters - who face intense wildfire smoke exposure - show persistent elevations in inflammatory markers for weeks after exposure ends. The chronic inflammation creates a feedback loop where damaged tissue releases more inflammatory signals, keeping the immune system on high alert.
This chronicity explains why patients often experience flares days or even weeks after smoke exposure. The immune system has a memory, and repeated exposures train it to react more aggressively each time. Medium-term exposure studies link cumulative wildfire smoke exposure to increased cardiorespiratory hospitalization risks, suggesting that even moderate exposures add up over time.
The dose-response relationship is clear: higher particulate concentrations and longer exposure durations lead to more severe and prolonged flares. But there's no truly "safe" threshold for individuals with autoimmune conditions - even brief exposures during moderate air quality days can trigger responses in highly sensitive patients.
Given the growing frequency of wildfire smoke events, autoimmune patients need practical, evidence-based protection strategies. The good news is that even modest interventions can significantly reduce exposure and flare risk.
Indoor air management: High-efficiency particulate air (HEPA) filters are the gold standard for indoor protection. Studies show that properly sized HEPA filters can remove over 99% of PM2.5 from indoor air. Place them in bedrooms and main living areas, and run them continuously during smoke events. Seal windows and doors with weather stripping, and avoid using window fans that pull outdoor air inside. Set air conditioning to recirculate mode rather than bringing in fresh air.

Personal protective equipment: Not all masks are created equal. Cloth masks and surgical masks offer minimal protection against PM2.5. N95 or KN95 respirators, fitted properly, can filter out 95% of fine particles. Respiratory protection is critical during outdoor activities, but fit matters - gaps around the nose or chin defeat the purpose. For patients with breathing difficulties, consult a healthcare provider about powered air-purifying respirators (PAPRs).
Timing and activity modification: Monitor air quality indexes (AQI) religiously through apps like AirNow or PurpleAir. When AQI exceeds 100 (unhealthy for sensitive groups), minimize outdoor time. Schedule essential activities for early morning when particulate levels are typically lowest. Avoid outdoor exercise entirely when AQI tops 150 - the increased respiratory rate during exercise dramatically increases particulate intake.
Anti-inflammatory support: While not a substitute for avoidance, certain interventions may help mitigate inflammatory responses. Antioxidant-rich diets (berries, leafy greens, omega-3 fatty acids) can bolster cellular defenses against oxidative stress. Stay well-hydrated to support mucosal clearance of particles. Some physicians recommend temporarily increasing anti-inflammatory medications during high-risk periods, though this requires individualized medical guidance.
Communication with healthcare providers: Alert your rheumatologist, immunologist, or primary care doctor at the start of wildfire season. Discuss contingency plans for flare management, including rescue medication protocols and when to seek urgent care. Track your symptoms in relation to air quality data - this documentation can help optimize your treatment plan and validate the connection between exposures and flares.
Individual actions matter, but the scale of the wildfire smoke crisis demands systemic solutions. Communities and policymakers must recognize autoimmune patients as a vulnerable population requiring targeted protections.
Public health infrastructure: Cities should establish clean air shelters - publicly accessible spaces with HEPA filtration where vulnerable residents can spend time during smoke events. Libraries, community centers, and shopping malls can serve this function if properly equipped and publicized. Subsidized or free HEPA filter distribution programs for low-income households and high-risk individuals would dramatically improve health equity.
Healthcare system preparedness: Emergency departments and urgent care clinics need protocols for identifying and treating smoke-related autoimmune flares. Many patients and even clinicians don't connect worsening symptoms to environmental exposures. Medical education should emphasize environmental triggers, and electronic health records could flag high-risk patients during smoke events for proactive outreach.
Land management and prevention: While beyond the scope of individual action, forest management practices that reduce catastrophic wildfire risk - controlled burns, fuel reduction, defensible space requirements - ultimately protect public health. Climate adaptation strategies must account for health impacts, not just property protection.
Research investment: Despite clear epidemiological links, we still lack definitive data on which autoimmune conditions are most affected, optimal protective strategies, and potential therapeutic interventions. Federal research funding should prioritize understanding immune-environment interactions and developing treatments that could block particulate-induced immune activation.
As climate change extends fire seasons and increases fire intensity, wildfire smoke will become an inescapable feature of life for millions. For the estimated 24 million Americans living with autoimmune conditions, this represents a profound challenge - but not an insurmountable one.
The emerging science reveals that wildfire smoke doesn't just irritate lungs; it fundamentally alters immune system behavior through multiple molecular pathways. PM2.5 particles carrying PAHs activate danger sensors, trigger inflammasomes, and promote B-cell hyperactivity - all mechanisms that can tip the balance toward autoimmune flares. The epidemiological evidence confirms what patients have long suspected: smoke exposure increases their disease burden measurably and meaningfully.
Yet knowledge is power. Armed with understanding of the mechanisms, timelines, and protective strategies, autoimmune patients can reduce their risk substantially. The combination of personal vigilance, medical partnership, and community support creates a robust defense against this environmental threat.
The path forward requires recognizing autoimmune patients as a population uniquely vulnerable to wildfire smoke and building systems to protect them. It means rethinking air quality guidelines to account for immune impacts, not just respiratory ones. It demands investment in research to unlock therapeutic approaches that could prevent smoke-triggered flares entirely.
We can't stop wildfires, and we can't immediately reverse climate change. But we can protect those most vulnerable to the immune chaos that wildfire smoke unleashes. The science now clearly illuminates the path - the question is whether we'll follow it before the next plume rolls in.

Saturn's moon Titan may harbour liquid water beneath its frozen crust, kept from freezing by ammonia acting as a natural antifreeze. New Cassini data suggests the interior could be slush with warm water pockets rather than a global ocean, and NASA's Dragonfly mission launching in 2028 aims to investigate whether this exotic environment could support life.

Bacteroides thetaiotaomicron uses 88 specialized gene clusters and over 260 enzymes to decode and digest dietary fibers humans can't break down, converting them into essential short-chain fatty acids. When fiber runs out, it eats your gut's protective mucus instead, with cascading health consequences.

Scientists are restoring Ice Age ecological dynamics through rewilding projects like Siberia's Pleistocene Park and de-extinction efforts by Colossal Biosciences. These initiatives aim to reintroduce megafauna or their proxies to repair broken ecosystems, protect Arctic permafrost, and slow climate change.

The cheerleader effect is a proven cognitive bias where people look more attractive in groups because the brain automatically averages faces, smoothing out individual flaws. Research shows the sweet spot is 3-5 people, it works for all genders, and it has real implications for dating apps and social media strategy.

The Hawaiian bobtail squid farms bioluminescent bacteria in a specialized light organ to erase its shadow in moonlit waters. This partnership, where bacteria reshape the squid's body and communicate through quorum sensing, is teaching scientists how host-microbe relationships work and inspiring new medical and biotech applications.

Millions are leaving social media platforms driven by privacy scandals, mental health concerns, and algorithmic manipulation. While 60% relapse within a week, those who stay away report dramatically improved wellbeing, and decentralized alternatives like Bluesky are surging.

Building a reliable quantum computer requires roughly 1,000 fragile physical qubits per logical qubit due to surface code error correction overhead. New code families like LDPC and neutral-atom platforms are racing to slash that ratio, with some teams claiming it could drop to as few as 5-to-1.